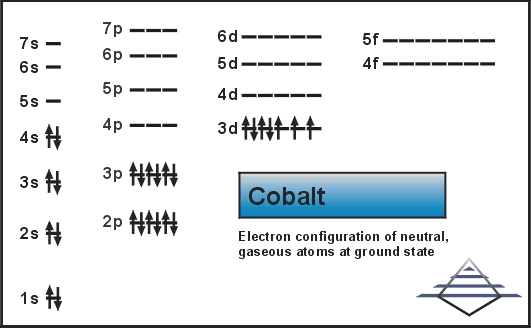
These elements are very chemically stable. The atoms along the right edge of the periodic table are called noble gases. Learn shorthand for writing long electron configurations. For instance, the first row of the d orbital block corresponds to the 3d orbital even though it's in period 4, while the first row of the f orbital corresponds to the 4f orbital even though it's in period 6. Caution: the d and f orbital regions of the table correspond to energy levels that are different from the period they're located in.Thus, its electron configuration will end. It's also in the fifth column of the periodic table's p orbital block. For example, when writing an electron configuration for Chlorine, think: "This atom is in third row (or "period") of the periodic table.Specifically, the 2 leftmost columns represent atoms whose electron configurations end in s orbitals, the right block of the table represents atoms whose configurations end in p orbitals, the middle portion, atoms that end in d orbital, and the bottom portion, atoms that end in f orbitals.Use the periodic table as a visual guide to write configurations – the order that you add electrons to orbitals corresponds to your position in the table. For example, atoms in the second column from the left always end in "s 2", atoms at the far right of the skinny middle portion always end in "d 10," etc. You may have already noticed that the shape of the periodic table corresponds to the order of orbital sets in electron configurations. Use the periodic table as a visual shortcut. Note that the above list, if all the shells were filled, would be the electron configuration for Og (Oganesson), 118, the highest-numbered atom on the periodic table-so this electron configuration contains every currently known electron shell for a neutrally charged atom.An electron configuration for an atom with every orbital completely filled would be written: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2 4f 14 5d 10 6p 6 7s 2 5f 14 6d 107p 6.Once you know the order of orbitals, you can simply fill them according to the number of electrons in the atom. For instance, a filled 4s 2 is lower energy (or less potentially volatile) than a partially-filled or filled 3d 10, so the 4s shell is listed first. Note that orbital sets are numbered by electron shell, but ordered in terms of energy. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
